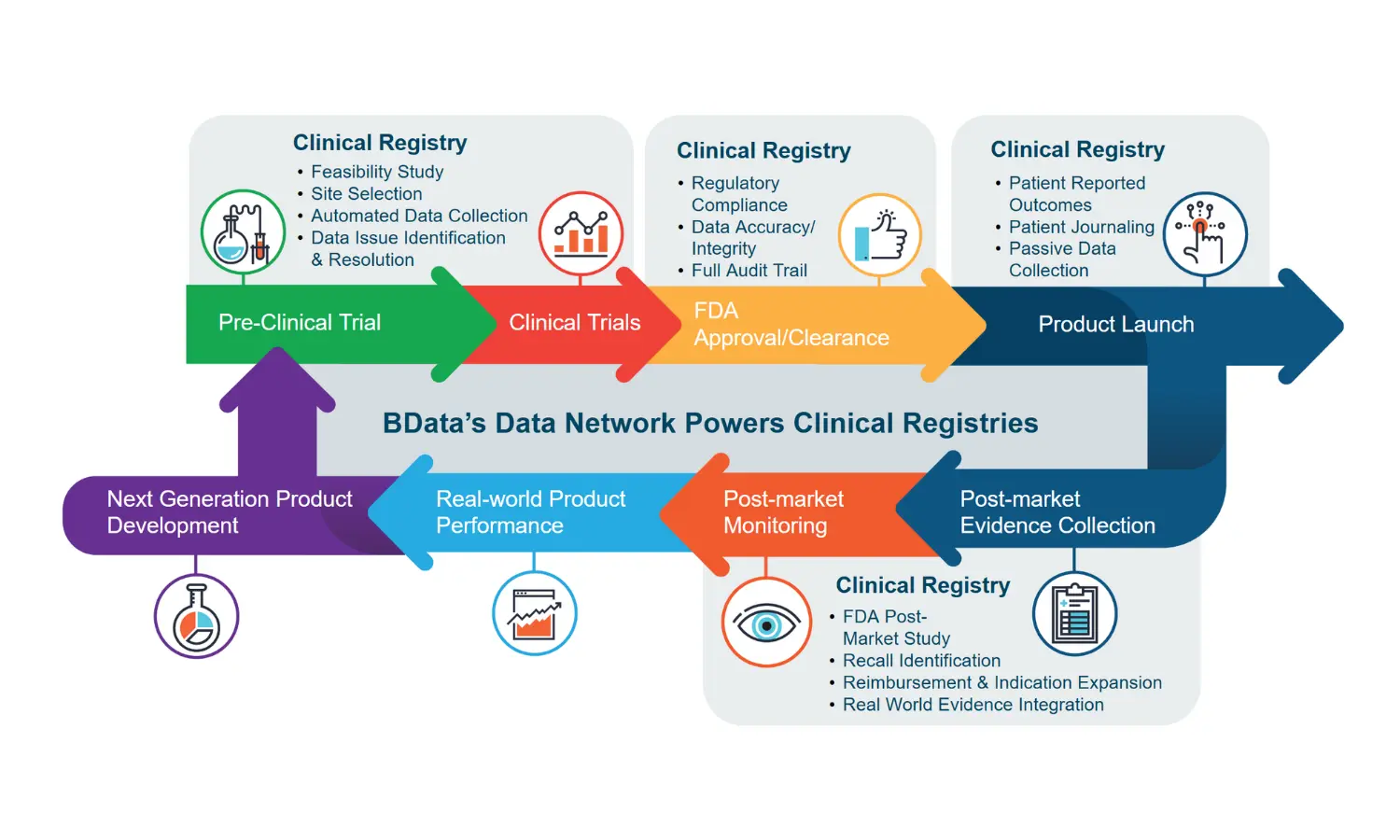
Get in-depth data on product utilization by category and key clinical treatment KPIs designed to help understand treatment successes and opportunities.
Leverage custom data sets from our proprietary BCQP to develop peer-reviewed publications to achieve post-market objectives. BData will partner with you to identify the exact data you need and create custom data sets to help you meet your objectives.
Leverage the BCQP’s proprietary, case-level real-world evidence (RWE), collected in collaboration with burn center clinicians and the American Burn Association (ABA), to support FDA's Total Product Life Cycle (TPLC) approach for generating clinical evidence for regulatory decision making.
Leveraging BCQP insights, BData partners with you to proactively identify study site risks and safeguards, monitor data collection and align study documents (e.g., protocol, monitoring plan, statistical analysis plan) with clinical practice.
We combine Real World Data (RWD) with prospective elements—to support patient replacement, augmentation, and comparator arm development—accelerating timelines, without sacrificing rigor.